Genetic predisposition of Stevens-Johnson Syndrome with Severe ocular surface complications
Stevens-Johnson syndrome (SJS) is an acute inflammatory vesiculobullous reaction of the skin and mucosa, such as the ocular surface, oral cavity, and genitals. In patients with extensive skin detachment and a poor prognosis, the condition is called toxic epidermal necrolysis (TEN). In the acute stage, patients manifest vesiculobullous lesions of the skin and mucosa, severe conjunctivitis, and ocular surface epithelial defects. In the chronic stage, despite the healing of the skin lesions, severe ocular surface complications persist such as severe dry eye, symblepharon, trichiasis, conjunctival invasion into the cornea and sometimes result in visual disturbance.
This proceeding consists of three subject, first subject is“Position of ophthalmic SJS”, second subject is “HLA analysis”, third subject is “Genome wide association study”, and forth subject is “other findings”.
1. Position of ophthalmic SJS
Diagnostic criteria for SJS in Japan 1.
Major Criteria consist of three findings; 1. Severe, hyperemic and/or hemorrhagic mucocutaneous lesions, 2. Epidermal detachment involves less than 10% of the total body surface area, 3. High fever (>38.0℃). The definite diagnosis of SJS need these all 3 major findings, which include mucosal lesions
Diagnostic criteria for TEN in Japan 1.
Major Criteria consist of three fondings; 1. Epidermal detachment involves more than 10% of the total body surface area, 2. Exclusion of staphylococcal scalded skin syndrome, 3. High fever (>38.0℃). The definite diagnosis of TEN need these all 3 major findings but they do not include mucosal lesions. So, some, not all, of TEN might have mucosal lesions.
Thus, SJS/TEN with mucosal lesion consist of SJS and a part of TEN. Moreover, not all, but some of SJS/TEN with mucosal lesions complicate severe ocular lesion such as severe conjunctivitis with psedomembrane and ocular surface epithelial defects.
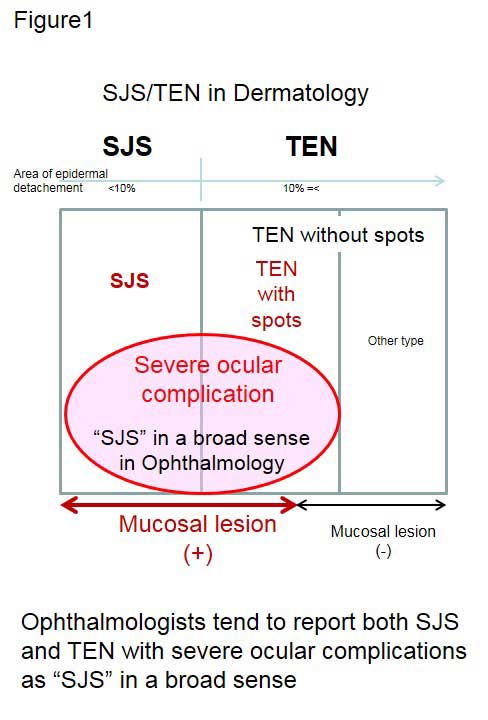
Furthermore, dermatologists see patients only in their acute stage. On the other hand, ophthalmologists encounter SJS/TEN patients not only in the acute stage but also in the chronic stage. So, it is difficult for ophthalmologists to render a differential diagnosis of SJS or TEN when patients present in the chronic stage because the vesiculobullous skin lesion expressed in the acute stage have healed by the chronic stage. Thus, ophthalmologists tend to report both SJS and TEN with severe ocular complications as “SJS” in a broad sense (Fig.1) 2. Our diagnosis of SJS/TEN (SJS in the broad sense) was based on a confirmed history of acute-onset high fever, serious mucocutaneous illness with skin eruptions, and involvement of at least 2 mucosal sites including the ocular surface.
Short summary is “Ophthalmic lesions appear in not all, but some of SJS/TEN patients who were diagnosed by dermatologists.”
2. HLA analysis
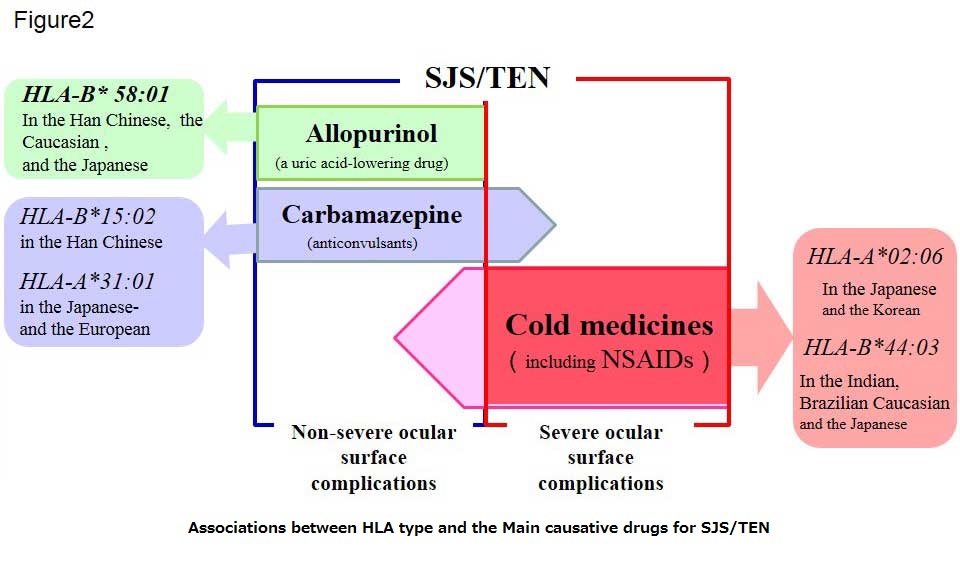
SJS/TEN are rare, but often associated with inciting drugs. Some causative drugs were reported, and some associations between HLA type and the drugs have also been reported. SJS/TEN induced by Allopurinol, a uric acid-lowering drug, was strongly associated with HLA-B* 58:01 in Han Chinese 3, Caucasian 4, and Japanese patients 5(Fig. 2). SJS/TEN induced by Carbamazepine, an anticonvulsant, was strongly associated with HLA-B*15:02 in Han Chinese 6, and HLA-A*31:01 in European 7 and Japanese 8(Fig. 2).
We have reported that cold medicines including NSAIDs and multi-ingredient cold medications were the main causative drugs for SJS/TEN with severe ocular complications 9, 10. About 80% of our patients have developed SJS/TEN after treatment for the common cold 10. Interestingly, allopurinol induces SJS/TEN without severe ocular complications (in submission). Moreover, not all cases of Carbamazepine-induced SJS/TEN have severe ocular complications 11. We performed HLA analysis of cold medicine related SJS/TEN with severe ocular complications (CM-SJS/TEN with SOC).
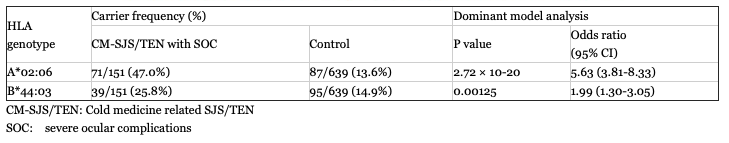
In Japanese, HLA-A*02:06 was strongly associated with CM-SJS/TEN with SOC, and HLA-B*44:03 also significantly associated with them (Table 1) 9.
Furthermore, compared to the Japanese CM-SJS/TEN without SOC, the significants remain 9. This result showed that HLA-A*02:06 is strongly associated with the Japanese CM-SJS/TEN with SOC, but not CM-SJS/TEN without SOC.
On the other hands, HLA-A*02:06 and HLA-B*44:03 are not associated with cold medicine unrelated (other medicine related) SJS/TEN with severe ocular surface complications9.
We also examined the association with HLA-A*02:06 and HLA-B*44:03 using other ethnic samples such as Korean, Indian and Brazilian samples. HLA-A*02:06 is also associated with Korean CM-SJS/TEN with SOC, but not Indian and Brazilian (Table2) 12. HLA-B*44:03 is associated with CM-SJS/TEN with SOC in Indian and Brazilian, but not Korean (Table2) 12. Especially in Indian, HLA-B*44:03 is strongly associated, 60% of the patients have HLA-B*44:03, while only 11% of controls have it, and odd’s ratio is 12. In Brazilian, when we focused only brazilian caucasian, 40% of the patients have HLA-B*44:03, while 11% of controls have it, odd’s ration is 6 12.
< TABLE 1 >
< TABLE 2 >
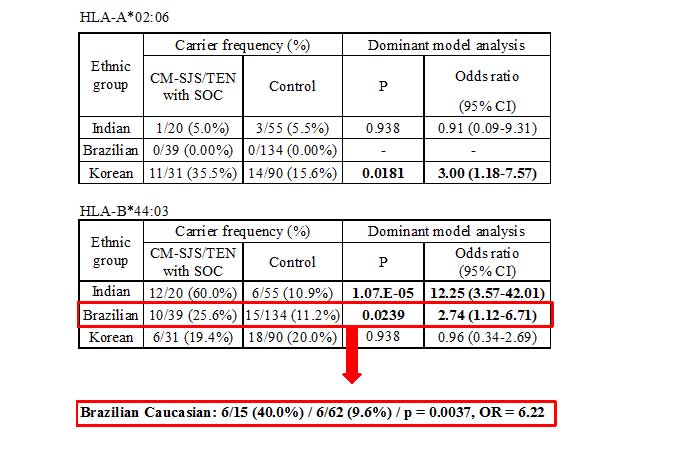
Short summary is “CM-SJS/TEN with SOC was associated with HLA-A*02:06 in Japanese and Korean and HLA-B*44:03 in Japanese, Indian and Brazilian Caucasian.” (Fig. 2).
3. Genome wide association study
We performed a genome-wide association study (GWAS) of CM-SJS/TEN with SOC.
We first enrolled Japanese 117 cases and 691 controls. We found that the HLA-A region showed the strongest association with susceptibility to CM-SJS/TEN with SOC. Outside of the HLA region, there were 57 SNPs with p < 10-3 (44 genes) in the allele frequency, and 9 SNPs of them (8 genes) were p < 10-5 13.
We selected 8 SNPs of 6 genes, for which functional TaqMan probe available, for the first replication study. There are 9 SNPs p< 10-5 in GWAS using Japanese samples (KPUM samples), for which functional TaqMan probe available 13. So, we focused the IKZF1 SNPs, we genotyped additional two SNPs of the gene (total 4 SNPs), which were included in the 57 SNPs with p < 10-3 outside of the HLA region in the GWAS 13. In the Korean samples, the 3 SNPs of IKZF1 were significantly associated, in the Indian samples, the 1 SNPs of IKZF1 were significantly associated 13. In the Brazillian samples, there were no significant associations with these IKZF1 SNPs; however, the ORs for these four SNPs with the Brazilian sample set showed the same direction of association as with Japanese samples13. Furthermore, the meta-analysis with Japanese, Korean, Indian and Brazillian samples showed a genome-wide significant association between CM-SJS/TEN with SOC and the IKZF1 SNP (rs4917014 (G vs T), OR = 0.5, p = 8.5×10-11, rs4917129 (C vs T), OR = 0.5, p = 8.1×10-9, rs10276619 (G vs A), OR = 1.8, p = 4.3×10-9) 13. These findings show that IKZF1 may be a universal marker for susceptibility to CM-SJS/TEN with SMI.
Finally, we examined the quantitative ratios of IKZF1 alternative splicing isoforms as functional analysis, and found that the ratio of Ik2/Ik1 was significantly associated with the SNPs of IKZF1 13.These results indicate that the ratio of Ik2/Ik1 may be influenced by IKZF1 SNP.”
Short summary is “We conducted a GWAS for CM-SJS/TEN with SMI in Japanese individuals and performed a replication study with Korean and Indian individuals, and found that IKZF1 SNPs were significantly associated with CM-SJS/TEN with SMI, and that the ratio of Ik2/Ik1 isoform may be influenced by these IKZF1 SNPs.”
4. Other finding
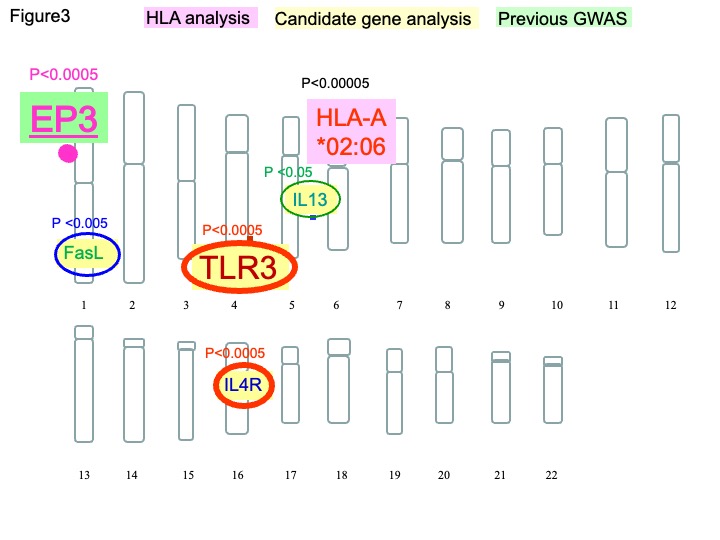
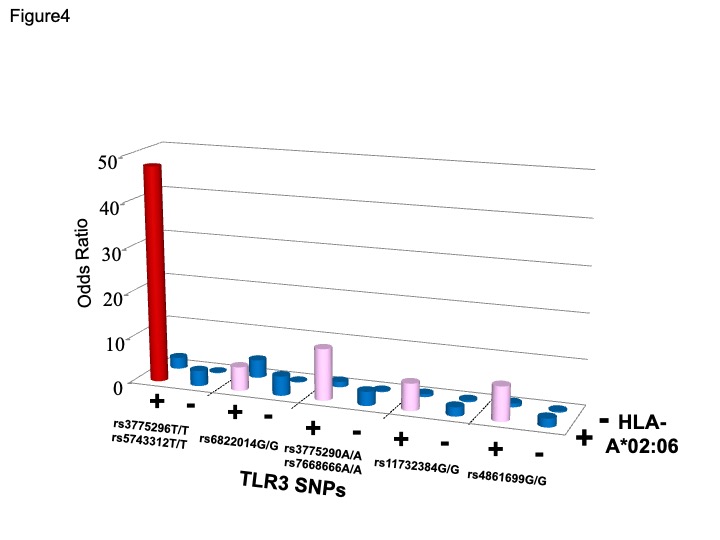
For the past decade, SNPs have been widely used as genetic markers for identifying human disease-susceptibility genes. However, it has become apparent that gene-gene interactions should be considered in addition to major single-locus effects 14. We previously reported that in addition HLA-A*02:06, TLR3 14-16, PTGER3 10, 14, IL-4R 17, 18, FasL 19 and IL-13 18 SNPs showed significant associations with SJS/TEN with severe ocular surface complications (Fig.3) and that HLA-A*02:06 with TLR3 polymorphisms exerted more than additive effects in SJS/TEN with SOC 16 (Fig.4).
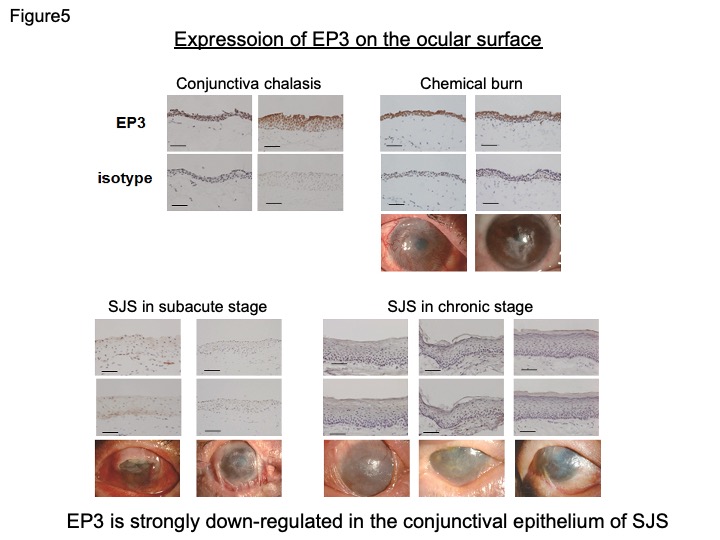
Furthermore, we also compared EP3 (protein of PTGER3 gene)expression in SJS and chemical burn. The chemical-burn conjunctival epithelium expressed the EP3 protein, the same as in conjunctivochalasis. However, we could not detect EP3 immunoreactivity in the conjunctival epithelium obtained from SJS, in either the chronic and sub-acute stages (Fig. 5) 20. These findings suggest that EP3 is strongly down-regulated in the conjunctival epithelium of SJS.
Cold medicines including NSAIDs (e.g., ibuprofen and loxoprofen) and cold medicine ingredients (e.g., acetaminophen), which were the main causative drugs for SJS/TEN with severe ocular complications, have the shared effect of down-regulating the production of prostanoid, including PGE2. Because the PGE2-EP3 pathway suppresses inflammation of the ocular surface 21, skin 22 and respiratory tract 23, we suggest that down-regulation of PGE2 by NSAIDs or acetaminophen might deeply contribute to the onset of CM-SJS/TEN with SOC.
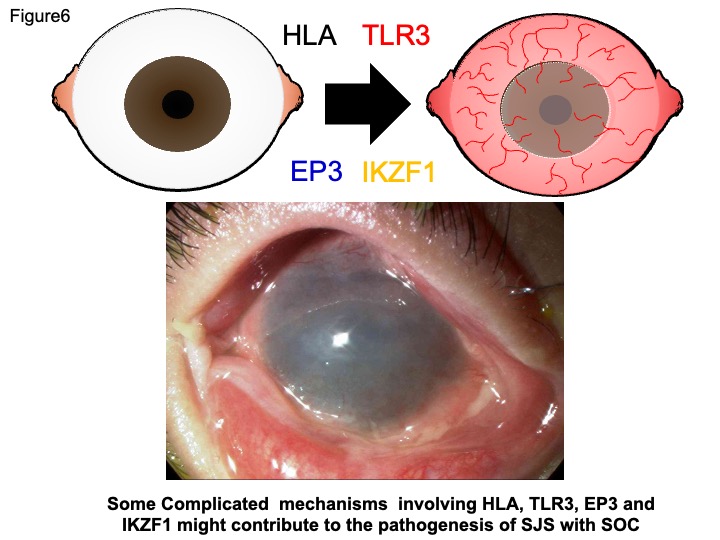
Thus, some complicated mechanisms involving HLA-A*02:06, TLR3, EP3 and IKZF1 might contribute the pathogenesis of SJS with SOC (Fig.6).
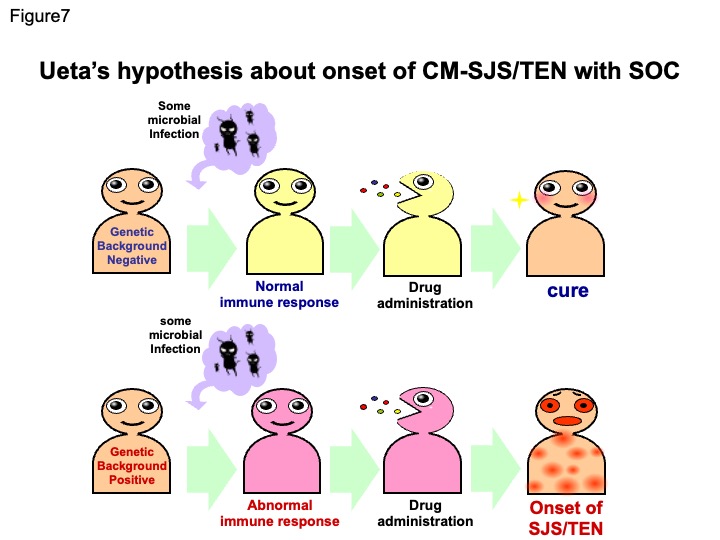
Moreover, CM-SJS/TEN patients have developed SJS/TEN by taking cold medicines after having a common cold as a result of some viral or mycoplasma infections. We postulate that not only cold medicine, but also an infectious agent, virus or other microbe, could together be important and necessary to trigger the onset of SJS/TEN with SOC. Ueta’s hypothesis about onset of SJS/TEN with SOC is that If a person has no genetic background of SJS/TEN with SOC when some microbial infection happens, he/she has normal immune responses, then takes cold medicine, resulting in cure of the common cold, but If a person has genetic background of SJS/TEN with SOC when some microbial infection happens, he/she has abnormal immune responses, then takes cold medicine, which triggers the onset of SJS/TEN with SOC (Fig.7).
References
- Sotozono C, Ueta M, Nakatani E, Kitami A, Watanabe H, Sueki H, et al. Predictive Factors associated with Acute Ocular Involvement in Stevens-Johnson Syndrome and Toxic Epidermal Necrolysis. Am J Ophthalmol in press.
- Ueta M, Kinoshita S. Ocular surface inflammation is regulated by innate immunity. Prog Retin Eye Res 2012; 31:551-75.
- Hung SI, Chung WH, Liou LB, Chu CC, Lin M, Huang HP, et al. HLA-B*5801 allele as a genetic marker for severe cutaneous adverse reactions caused by allopurinol. Proc Natl Acad Sci U S A 2005; 102:4134-9.
- Lonjou C, Borot N, Sekula P, Ledger N, Thomas L, Halevy S, et al. A European study of HLA-B in Stevens-Johnson syndrome and toxic epidermal necrolysis related to five high-risk drugs. Pharmacogenet Genomics 2008; 18:99-107.
- Tohkin M, Kaniwa N, Saito Y, Sugiyama E, Kurose K, Nishikawa J, et al. A whole-genome association study of major determinants for allopurinol-related Stevens-Johnson syndrome and toxic epidermal necrolysis in Japanese patients. Pharmacogenomics J 2013; 13:60-9.
- Chung WH, Hung SI, Hong HS, Hsih MS, Yang LC, Ho HC, et al. Medical genetics: a marker for Stevens-Johnson syndrome. Nature 2004; 428:486.
- McCormack M, Alfirevic A, Bourgeois S, Farrell JJ, Kasperaviciute D, Carrington M, et al. HLA-A*3101 and carbamazepine-induced hypersensitivity reactions in Europeans. N Engl J Med 2011; 364:1134-43.
- Ozeki T, Mushiroda T, Yowang A, Takahashi A, Kubo M, Shirakata Y, et al. Genome-wide association study identifies HLA-A*3101 allele as a genetic risk factor for carbamazepine-induced cutaneous adverse drug reactions in Japanese population. Hum Mol Genet 2011; 20:1034-41.
- Ueta M, Kaniwa N, Sotozono C, Tokunaga K, Saito Y, Sawai H, et al. Independent strong association of HLA-A*02:06 and HLA-B*44:03 with cold medicine-related Stevens-Johnson syndrome with severe mucosal involvement. Sci Rep 2014; 4:4862.
- Ueta M, Sotozono C, Nakano M, Taniguchi T, Yagi T, Tokuda Y, et al. Association between prostaglandin E receptor 3 polymorphisms and Stevens-Johnson syndrome identified by means of a genome-wide association study. J Allergy Clin Immunol 2010; 126:1218-25 e10.
- Kaniwa N, Saito Y, Aihara M, Matsunaga K, Tohkin M, Kurose K, et al. HLA-B locus in Japanese patients with anti-epileptics and allopurinol-related Stevens-Johnson syndrome and toxic epidermal necrolysis. Pharmacogenomics 2008; 9:1617-22.
- Ueta M, Kannabiran C, Wakamatsu TH, Kim MK, Yoon KC, Seo KY, et al. Trans-ethnic study confirmed independent associations of HLA-A*02:06 and HLA-B*44:03 with cold medicine-related Stevens-Johnson syndrome with severe ocular surface complications. Sci Rep 2014; 4:5981.
- Ueta M, Sawai H, Sotozono C, Hitomi Y, Kaniwa N, Kim MK, et al. IKZF1, a new susceptibility gene for cold medicine-related Stevens-Johnson syndrome/toxic epidermal necrolysis with severe mucosal involvement. J Allergy Clin Immunol 2015.
- Ueta M, Tamiya G, Tokunaga K, Sotozono C, Ueki M, Sawai H, et al. Epistatic interaction between Toll-like receptor 3 (TLR3) and prostaglandin E receptor 3 (PTGER3) genes. J Allergy Clin Immunol 2012; 129:1413-6 e11.
- Ueta M, Sotozono C, Inatomi T, Kojima K, Tashiro K, Hamuro J, et al. Toll-like receptor 3 gene polymorphisms in Japanese patients with Stevens-Johnson syndrome. Br J Ophthalmol 2007; 91:962-5.
- Ueta M, Tokunaga K, Sotozono C, Sawai H, Tamiya G, Inatomi T, et al. HLA-A*0206 with TLR3 polymorphisms exerts more than additive effects in Stevens-Johnson syndrome with severe ocular surface complications. PLoS One 2012; 7:e43650.
- Ueta M, Sotozono C, Inatomi T, Kojima K, Hamuro J, Kinoshita S. Association of IL4R polymorphisms with Stevens-Johnson syndrome. J Allergy Clin Immunol 2007; 120:1457-9.
- Ueta M, Sotozono C, Inatomi T, Kojima K, Hamuro J, Kinoshita S. Association of combined IL-13/IL-4R signaling pathway gene polymorphism with Stevens-Johnson syndrome accompanied by ocular surface complications. Invest Ophthalmol Vis Sci 2008; 49:1809-13.
- Ueta M, Sotozono C, Inatomi T, Kojima K, Hamuro J, Kinoshita S. Association of Fas Ligand gene polymorphism with Stevens-Johnson syndrome. Br J Ophthalmol 2008; 92:989-91.
- Ueta M, Sotozono C, Yokoi N, Inatomi T, Kinoshita S. Prostaglandin E receptor subtype EP3 expression in human conjunctival epithelium and its changes in various ocular surface disorders. PLoS One 2011; 6:e25209.
- Ueta M, Matsuoka T, Narumiya S, Kinoshita S. Prostaglandin E receptor subtype EP3 in conjunctival epithelium regulates late-phase reaction of experimental allergic conjunctivitis. J Allergy Clin Immunol 2009; 123:466-71.
- Honda T, Matsuoka T, Ueta M, Kabashima K, Miyachi Y, Narumiya S. Prostaglandin E(2)-EP(3) signaling suppresses skin inflammation in murine contact hypersensitivity. J Allergy Clin Immunol 2009; 124:809-18 e2.
- Kunikata T, Yamane H, Segi E, Matsuoka T, Sugimoto Y, Tanaka S, et al. Suppression of allergic inflammation by the prostaglandin E receptor subtype EP3. Nat Immunol 2005; 6:524-31.



